Enantioselective Ni(0)-Catalyzed Annulation of Pyridones
Pyridones are common structural motifs in natural products exhibiting diverse biological activity and hence the pyridine core is found in a variety of pharmacologically potent compounds.[1] In particular 1,6-carboannulated pyridones are found in several biologically active natural products and furthermore annulated 2-pyridones can serve as access to valuable bioactive indolizidine and quinolizidine alkaloids.[2]
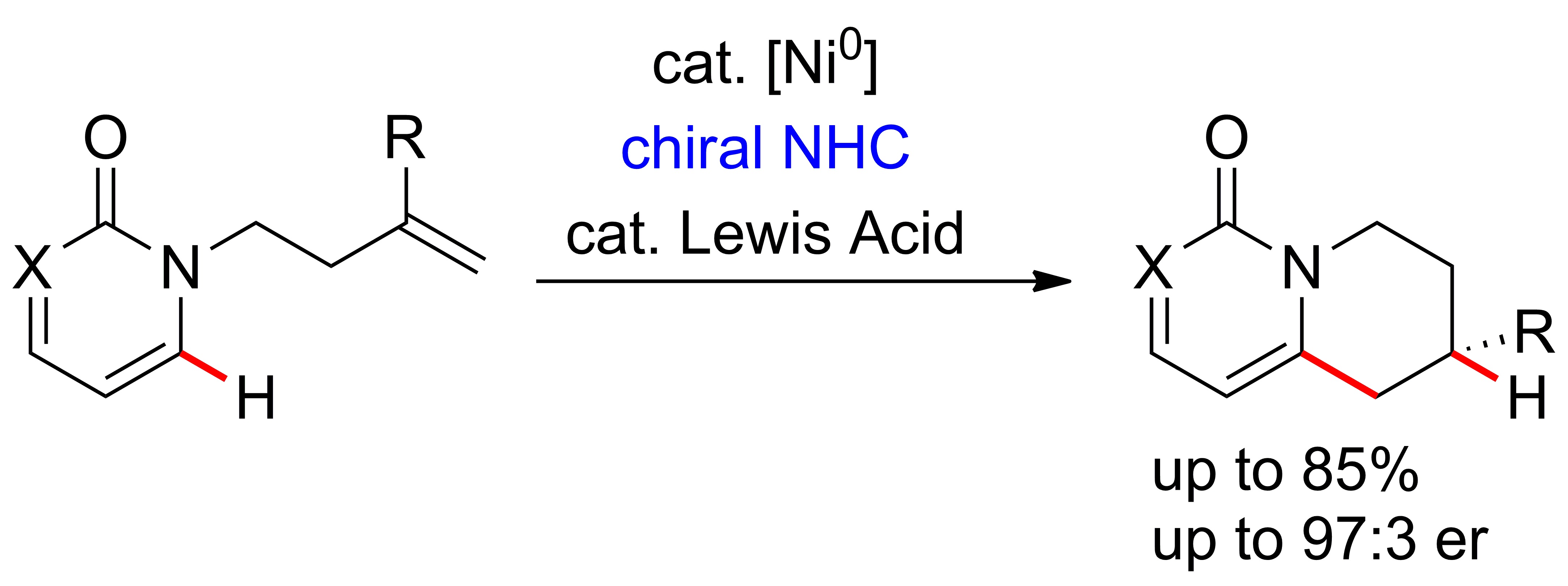
Based on the work of Nakao and Hiyama our group has developed a Nickel catalyzed endo-selective annulation protocol of N-alkenyl-2-pyridones.[3,4] Cooperative Lewis Acid/Ni(0)-catalysis and application of N-heterocyclic carbene ligands enabled C-H activation and subsequent regioselective cyclization under formation of a stereocenter. A variety of known chiral NHCs failed to achieve a highly enantioselective transformation, highlighting the need for further ligand development in this area.
We have developed a class of chiral NHCs which enable the formation of valuable 1,6-annulated 2‑pyridones in high enantioselectivity.
[1] Q. Li, A. Claiborne, T. Li, L. Hasvold, V. S. Stoll, S.Muchmore, C. G. Jakob, W. Gu, J. Cohen, C. Hutchins, D.Frost, S. H. Rosenberg, H. L. Sham, Bioorg. Med. Chem. Lett., 2004, 14, 5367–5370.
[2] M. E. Wall, M. C. Wani, C. E. Cook, K. H.Palmer, J. Am. Chem. Soc., 1966, 88, 3888–3890.
[3] a) Y. Nakao, H. Idei, K. S. Kanyiva, T. Hiyama, J. Am. Chem.Soc., 2009, 131, 15996–15997; b) R. Tamura, Y. Yamada, Y.Nakao, T. Hiyama, Angew. Chem. Int. Ed., 2012, 51, 5679–5682.
[4] P. A. Donets, N. Cramer, Angew. Chem. Int. Ed., 2015, 54, 633–637.