Synthesis of Vinyl Triazenes by Pd-Catalyzed Addition Reactions to 1-Alkynyltriazenes
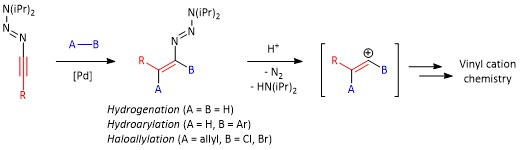
In 2015, our group reported a synthetic method for the preparation of 1-alkynyltriazenes using nitrous oxide (N2O), lithium N,N-dialkylamides and 1-alkynylmagnesiumbromides. [1] Subsequently, we have shown that 1-alkynyltriazenes have a similar reactivity profile as ynamides. [2] In this work, we have studied three Pd-catalyzed addition reactions to 1-alkynyltriazenes: semi-hydrogenation, hydroarylation with arylboronic acids, and haloallylation with allylhalides. Reactions have a broad scope and proceed with good to excellent yields and high regio- and stereoselectivity giving multisubstituted variety of vinyl triazenes. Treatment of vinyl triazenes with a strong acid leads to rapid formation of the corresponding vinyl cations, which can be captured by nucleophiles.
[1] G. Kiefer, T. Riedel, P. J. Dyson, R. Scopelliti, K. Severin Angew. Chem. Int. Ed. 2015, 54, 302.
[2] F. G. Perrin, G. Kiefer, L. Jeanbourquin, S. Racine, D. Perrotta, J. Waser, R. Scopelliti, K. Severin Angew. Chem. Int. Ed. 2015, 54, 13393.