Exploration of Pd(0)-Catalysed C(sp3)−H Functionalisation Beyond Aryl Halides
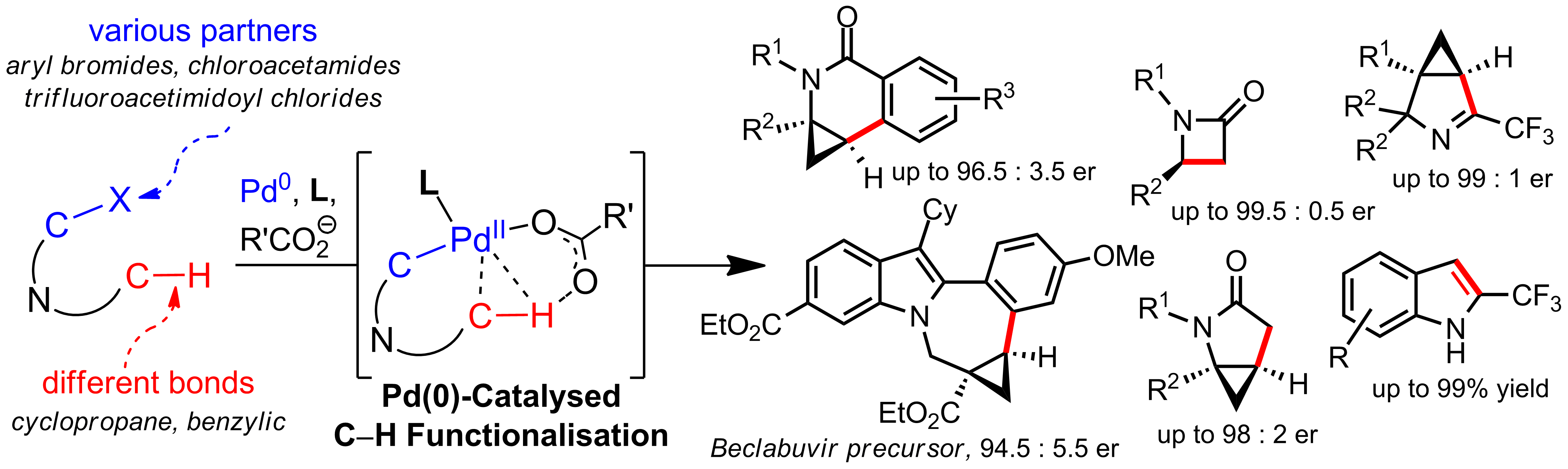
Nitrogen-containing heterocycles are prevalent motifs in biologically active compounds.1 Transition metal catalysed enantioselective C–H functionalisations have become attractive alternatives for the selective synthesis of such scaffolds.2 In the past years, the enantioselective synthesis of benzannulated N-heterocyclic building blocks via intramolecular Pd(0)‑catalysed C(sp3)–H bond arylation has been extensively investigated.3 In this context, we have developed intramolecular aminocyclopropane arylations towards dihydroisoquinolinones and the Beclabuvir ring system.4
Our recent studies broaden the scope of Pd(0)‑catalysed C–H functionalisations by using electrophilic partners other than aryl halides. Readily accessible chloroacetamides are efficiently functionalised, yielding valuable chiral b‑ and g‑lactams in high yields and enantioselectivities with formation of a C(sp3)-C(sp3) bond.5,6 Furthermore, indoles and versatile chiral imines bearing a CF3-group are obtained by C–H functionalisation of trifluoroacetimidoyl chlorides.7
[1] E. Vitaku, D. T. Smith, J. T. Njardarson, J. Med. Chem. 2014, 57, 10257
[2] C. G. Newton, S.-G. Wang, C. C. Oliveira, N. Cramer, Chem. Rev. 2017, DOI: 10.1021/acs.chemrev.6b00692
[3] O. Baudoin, Acc. Chem. Res. 2017, 50, 1114
[4] J. Pedroni, T. Saget, P. A. Donets, N. Cramer, Chem. Sci. 2015, 6, 5164-5171
[5] J. Pedroni, M. Boghi, T. Saget, N. Cramer, Angew. Chem. Int. Ed. 2014, 53, 9064
[6] J. Pedroni, N. Cramer, Angew. Chem. Int. Ed. 2015, 54, 11826
[7] J. Pedroni, N. Cramer, Org. Lett. 2016, 18, 1932