Nickel complexes containing oxygen-chelating mesoionic carbenes as cheap and efficient hydrosilylation catalysts.
Research into new and efficient catalytic systems based on Earth-abundant metals represents a significant challenge for chemistry.1 A key prerequisite to reach this goal is the availability of suitable ligands to stabilize the metal center. This field has been considerably stimulated by the discovery of N-heterocyclic carbenes (NHCs) as ligands that impart unique properties.2Triazolylidenes are a subclass of NHCs that are excellent σ-donors and have great electronic flexibility, which allows for the stabilization of a wide variety of oxidation states of transition metals. Furthermore, their convenient synthetic modification provides access to heteroatom-functionalized NHCs for chelation. These donor-functionalized ligands have great potential for tailoring the properties of Earth-abundant metals for catalysis.
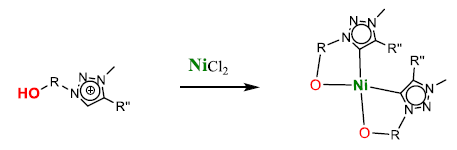
Here we will present a facile and versatile synthesis of mesoionic ligands bearing a potentially chelating hydroxide moiety on the triazole scaffold as well as a new class of nickel(II) complexes containing these O,C bidentate chelating mesoionic carbenes starting from a cheap and simple metal precursor. All these nickel complexes show high activity in hydrosilylation reactions, demonstrating the great potential of the hydroxyl group as chelating moiety for both the synthesis and the catalytic application of 3d metal complexes.
[1] R.M. Bullock, “Catalysis without precious metals”, 2011, Wiley-VCH, Weinheim, Germany.
[2] K.H. Dötz, J. Stendel, Chem. Rev. 2009, 109, 3227,F.E. Hahn, M. C.Jahnke, Angew. Chem. Int. Ed. Engl., 2008, 47, 3112, W.A Herrmann, Angew. Chem. Int. Ed., 2002, 41, 1290., J.D. Crowley, A. Lee, K. J. Kilpin, Aust. J. Chem., 2011, 64, 1118.