Aluminum Chloride–Natural Graphite Battery and its Energy Density
Non-aqueous, ionic liquid-based aluminum chloride-graphite batteries emerge as a highly promising post-Li-ion technology for low-cost and large-scale storage of electricity, because it features exclusively highly abundant chemical elements and simple fabrication methods. In this work, we examined the recently proposed aluminum–ionic liquid–graphite architecture.1 Although previous studies have focused on graphitic cathodes, we analyzed the practicality of achievable energy densities and found that the AlCl3-based ionic liquid is a capacity-limiting anode material. By focusing on both the graphitic cathode and the AlCl3-based anode, we improved the overall energy density.3,4 First, high cathodic capacities of ≤150 mAh g–1 and energy efficiencies of 90% at high electrode loadings of at least 10 mg cm–2 were obtained with highly crystalline natural graphite flakes or with synthetic kish graphite flakes, which were subjected to minimal mechanical processing. Second, the AlCl3 content in the ionic liquid was increased to its maximal value, which essentially doubled the energy density of the battery, resulting in a cell-level energy density of ≤65 Wh kg–1.
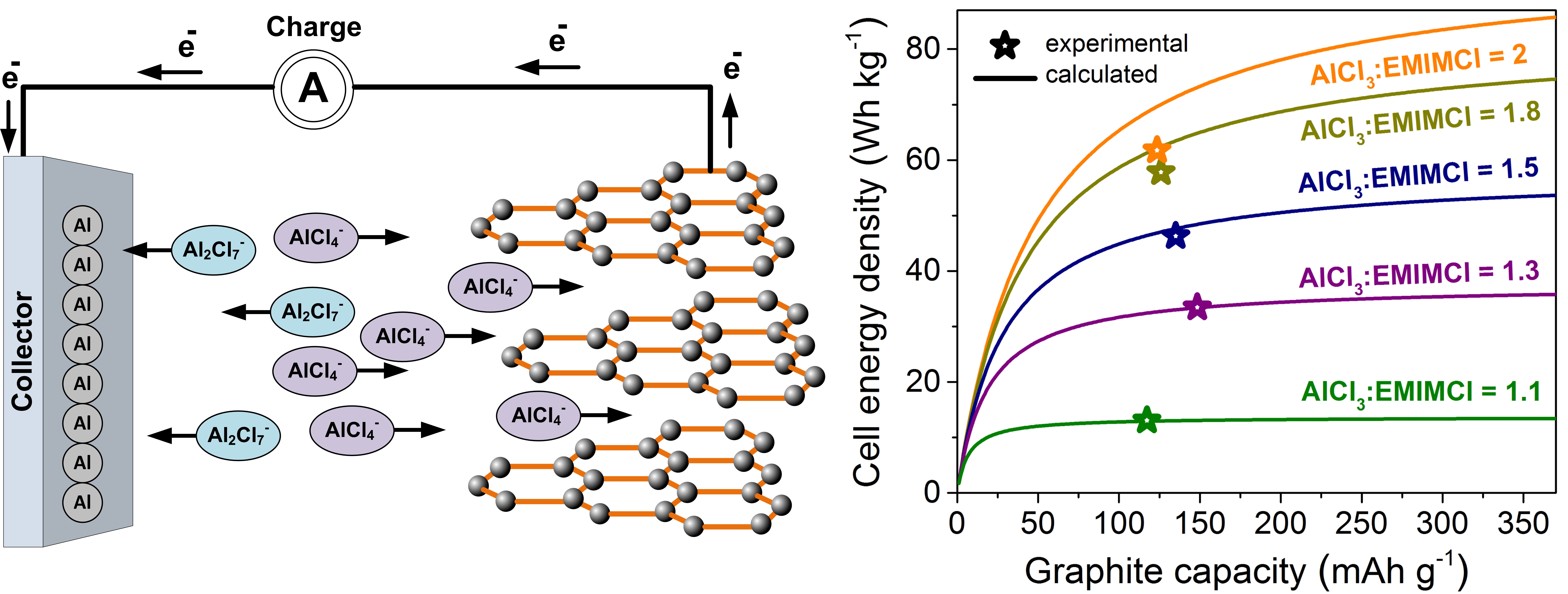
Figure 1. An aluminum chloride-graphite battery. (a) Schematics of the charging process. (b) Comparison of the calculated (curves) and experimental (data points) cell-level energy densities.
[1] Meng-Chang Lin, Ming Gong, Bingan Lu, Yingpeng Wu, Di-Yan Wang, Mingyun Guan, Michael Angell, Changxin Chen, Jiang Yang, Bing-Joe Hwang Hongjie Dai, Nature 2015, 520, 324-328.
[2] Kostiantyn V. Kravchyk, Shutao Wang, Laura Piveteau and Maksym V. Kovalenko, Chem. Mater. 2017, DOI: 10.1021/acs.chemmater.7b01060.
[3] Shutao Wang, Kostiantyn V. Kravchyk, Frank Krumeich and Maksym V. Kovalenko. Chem. Mater. 2017, submitted.