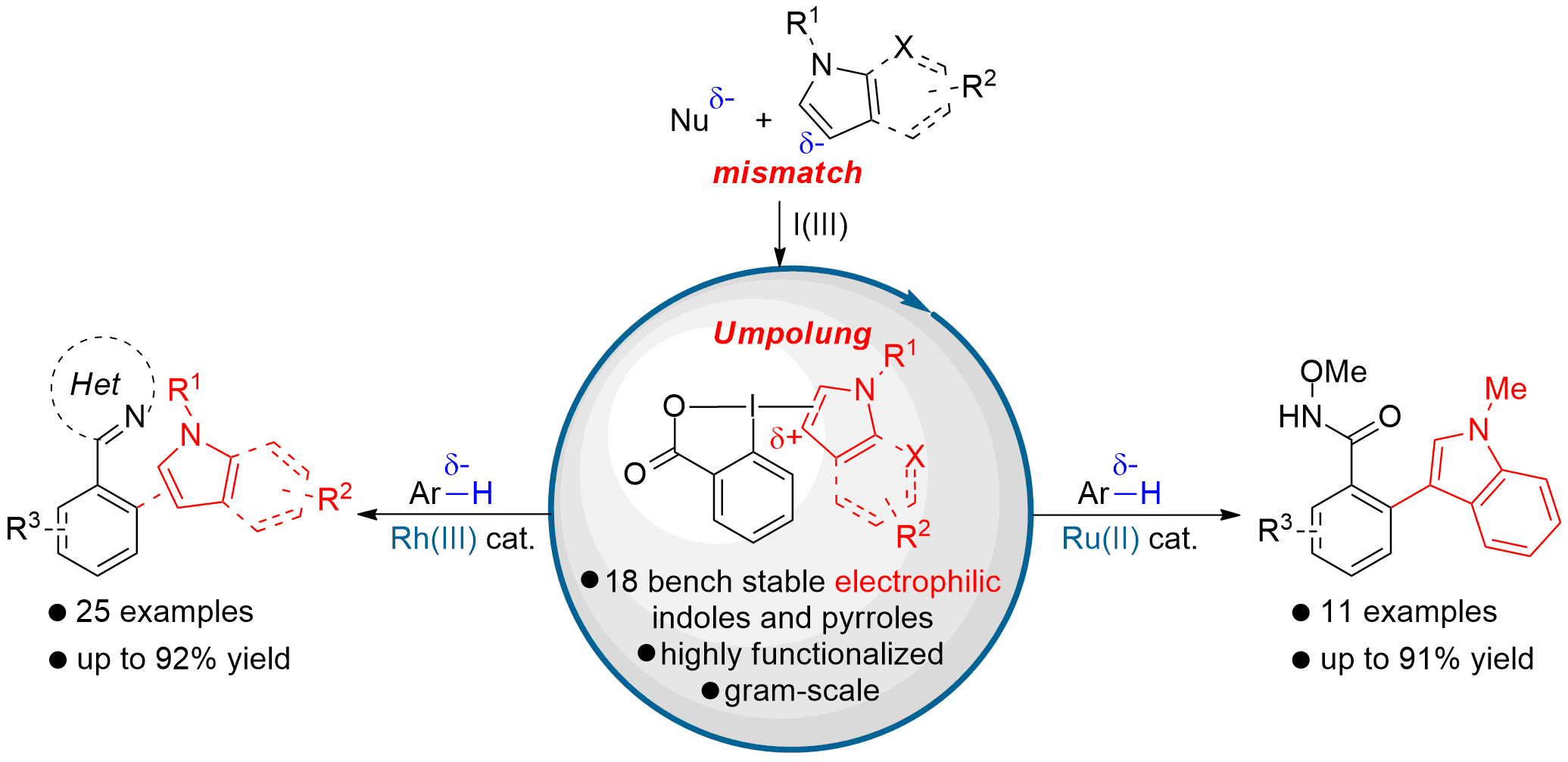
Bench-stable Electrophilic Indole and Pyrrole Benziodoxolone Reagents for C-H functionalization
Amongst all the azoles present in nature, electron-rich pyrroles and indoles are the most ubiquitous.[1] Their application as pharmaceuticals, fragrances and agrochemicals, as well as their relevance as natural bioactive compounds, led to the development of countless methods for both their synthesis[1] and functionalization.[2] While the nucleophilic properties of indoles and pyrroles have been intensively exploited, their “umpolung” into electrophilic synthons remained largely unexplored, although it would be of high interest for the development of new synthetic disconnections. Up to now, few groups reported the synthesis of unstable hypervalent indoles iodonium salts; however the number of applications was limited.[3] We present herein the first synthesis of 18 new bench stable electrophilic indole and pyrrole[4] benziodoxolone[5] reagents. Using these new reagents, rhodium(III) and ruthenium(II) catalyzed C-H “indolization” of nucleophilic arenes were developed to furnish the desired products with complete regioselectivity. The obtained functionalized indoles and pyrroles could not be synthesized using existing metal catalyzed C-H arylation processes and are expected to be useful intermediates for the synthesis of optoelectronical devices and bioactive compounds.
[1] Gordon W. Gribble. Indole Ring Synthesis: From Natural Products to Drug Discovery; Wiley.
[2] Bandini, M.; Eichholzer, A. Angew. Chem. Int. Ed. 2009, 48, 9608–9644.
[3] Neilands, O. Chem. Heterocycl. Compd. 2003, 39, 1555–1569.
[4] Caramenti, P.; Nicolai, S.; Waser, J. submitted.
[5] Li, Y.; Hari, D. P.; Vita, M. V.; Waser, J. Angew. Chem. Int. Ed. 2016, 55, 4436–4454.