Enhanced Catalytic Activity of Iridium(III) Complexes by Facile Modification of C,N-bidentate Chelating Pyridylideneamide Ligands
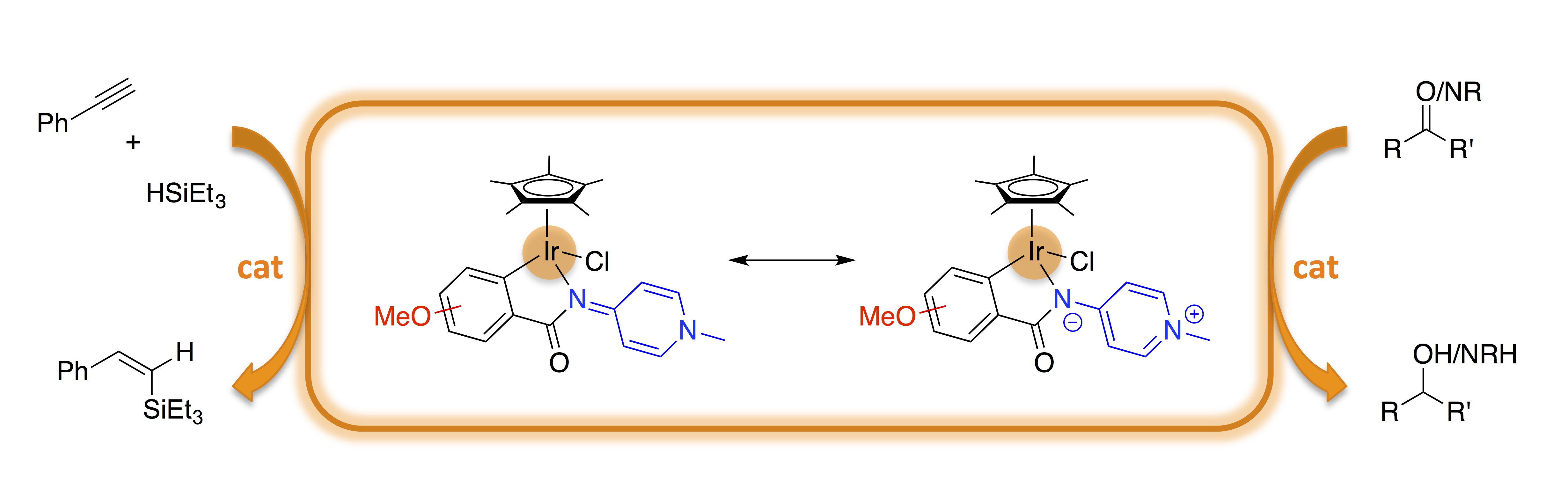
Non-innocent ligands that can significantly modulate their donor properties have become increasingly popular as a powerful class of ligands for a variety of homogenous catalytic applications, since they can stabilize different metal configurations.1 Pyridylideneamines (PYEs) and pyridylideneamides (PYAs) are electronically highly flexible N-donor sites that can coordinate to the metal center as a π-acidic imine or as a π-basic pyridinium amide. Their flexibility is represented by the two limiting resonance forms comprised of a diene heterocycle and a neutral imine donor site with minimal charge separation and a zwitterionic form, which features an anionic amide donor site and aromatic stabilization of the pyridinium residue.2
Here, we applied this concept for facile catalyst tailoring by incorporating donor substituents in different positions of the phenyl ring of the C,N-bidentate chelating PYA ligand (Fig. 1). These modifications greatly enhance the catalytic activity of the coordinated iridium center.
[1] Hartwig, J. Organotransition Metal Chemistry, University Science Books, Mill Hill Valley California, 2010.
[2] a) Donnelly, K. F.; Segarra, C.; Shao, L.; Suen, R.; Müller-Bunz, H.; Albrecht, M.; Organometallics 2015, 34, 4076−4084. b) Navarro, M.; Li, M.; Müller-Bunz, H.; Bernhard, S.; Albrecht, M.; Chem. Eur. J. 2016, 22, 6740–6745.