Toward a useful catalytic transformation of N2O using group 9 organometallic complexes
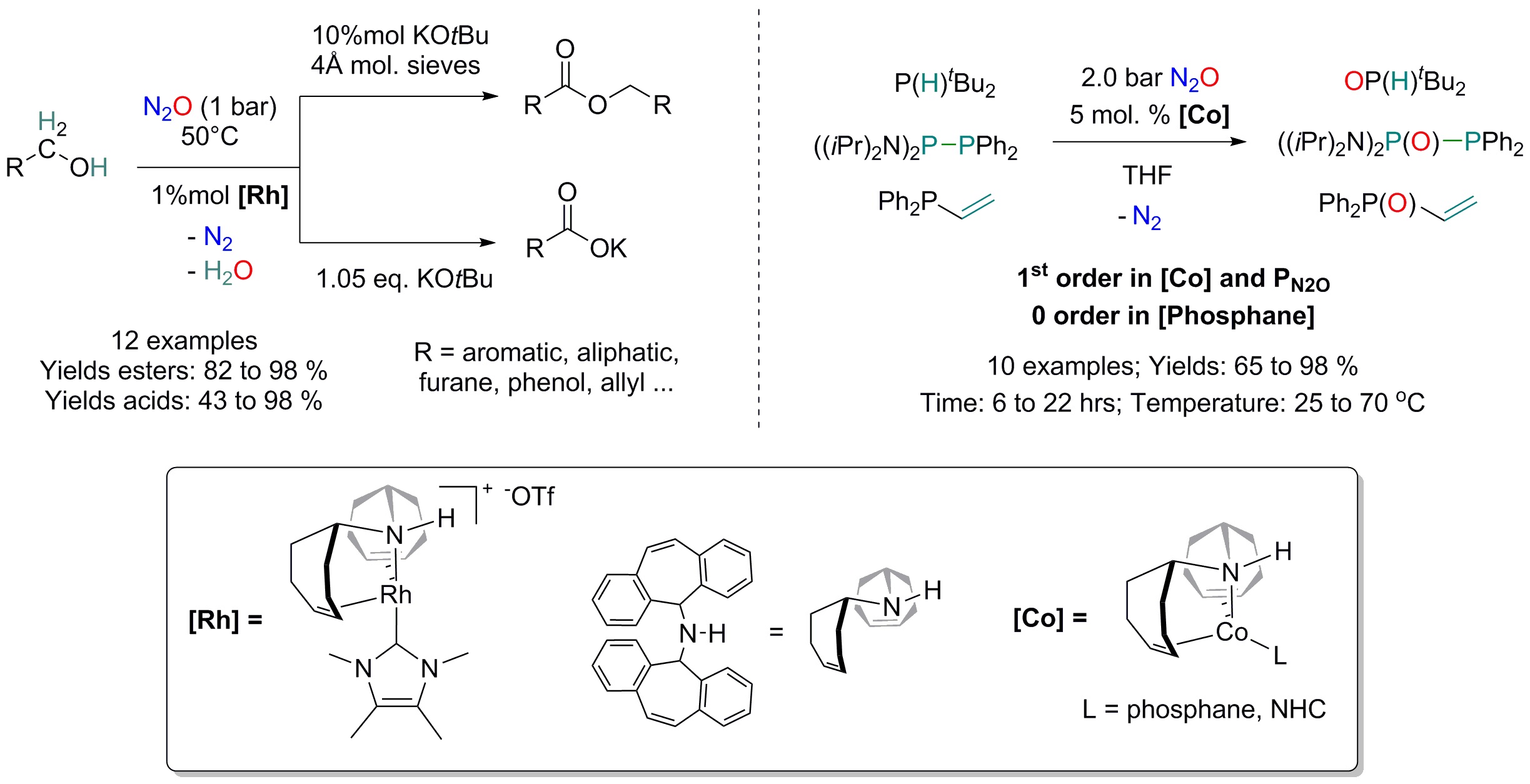
Nitrous oxide (N2O) gases have been recently identified as the largest global ozone depleting agents and as the 3rd largest emitted greenhouse gases worldwide and 300 times more powerful than CO2.[1] N2O is naturally produced via nitrification and denitrification of nitrate during nitrogen cycle, but is also an industrial waste. N2O emission has increased significantly during industrialization as a result of agricultural soil management, N-fertilizer use, livestock waste management, mobile & stationary fossil fuel, combustion and industrial processes. Its transformation to less harmful chemicals is of particular interest but very challenging, since even if thermodynamically unstable, nitrous oxide is kinetically inert.[2] We have successfully design low valent and reactive organometallic species containing group 9 metals (Rh[3] and Co[4]) that activate and catalytically transform, under mild conditions, this environmentally unfriendly molecules to valuable chemicals.
[1] a) A. R. Ravishankara, J. S. Daniel, R. W. Portmann, Science, 2009, 326, 123-125. b) J. Hansen, M. Sato, Proc. Natl. Acad. Sci. USA., 2004, 101, 16109-16114.
[2] a) E. Eger, I., II. In Nitrous Oxide N2O, Elsevier: New York, 1985. b) K. Severin Chem. Soc. Rev., 2015, 44, 6375-6386.
[3] T. L. Gianetti, S. P. Annen, G. Santiso-Quinones, M. Reiher, M. Driess, H. Grützmacher, Angew. Chem. Int. Ed., 2016, 55, 1886-1890.
[4] T. L. Gianetti, R. E. Rodriguez-Lugo, J. Harmer, M. Trincado, M. Vogt, G. Santiso-Quinones, H. Grützmacher, Angew. Chem. Int. Ed., 2016, 55, 15323-15328.