More than a solvent: donor-acceptor complexes of room-temperature ionic liquids and electron acceptors
Room-temperature Ionic liquids (RTILs) are considered as green alternatives to “conventional” solvents due to their low volatility, high intrinsic conductivity and good solubility for inorganic and organic solutes. Besides their applications, the elucidation of solvation and reaction dynamics in this unusual solvent is matter of interest. [1] The interaction of charged solvent molecules with solutes should impact the driving force, equilibrium and kinetics of elementary photochemical processes, especially those involving charged species such as electron or proton transfer reactions. The quantification of these effects, especially in bimolecular reactions, has turned out to be rather complex due to the high viscosity of RTILs which requires elaborate data analysis and may hide the impact of solvent-solute interactions on a molecular level. [2] Furthermore, it has to be questioned if the reactivity and stability of RTILs are sufficient to treat them as inert solvent if reactive intermediates are formed during e.g. photoinduced electron transfer.
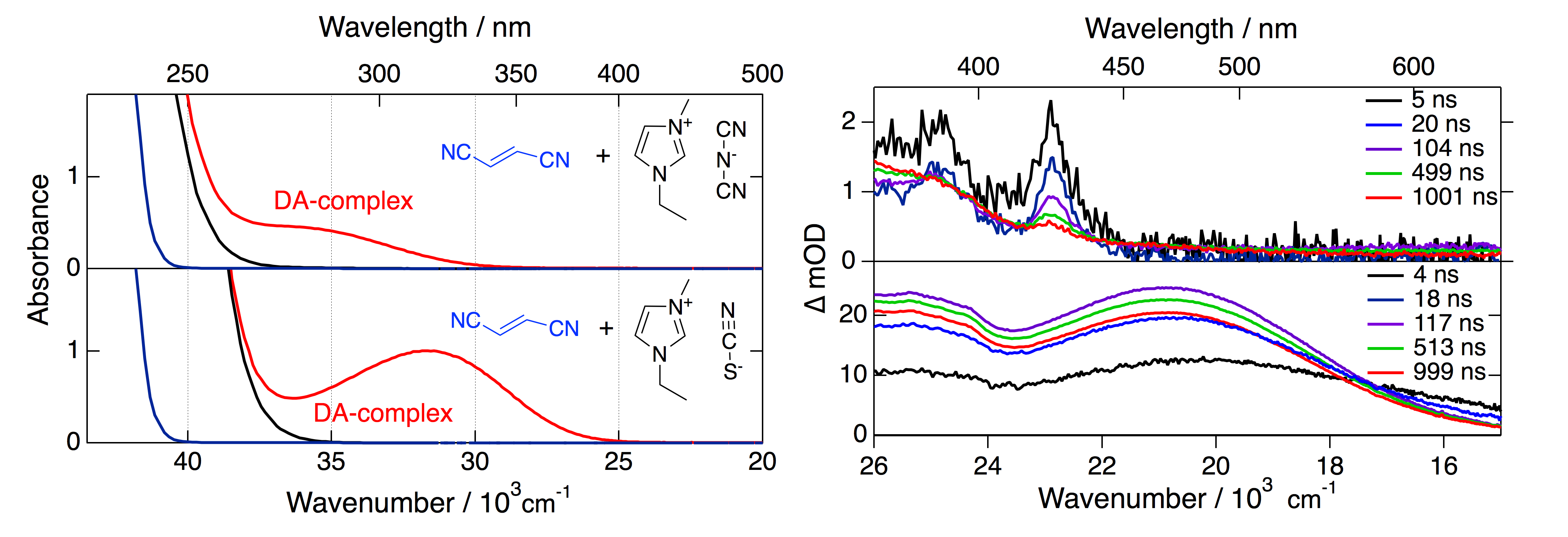
In our communication, we will show the appearance of broad charge-transfer (CT) absorption bands upon addition of electron donors (EDs) to solutions of RTILs in acetonitrile. These broad bands reveal the formation of ground-state donor-acceptor (DA) complexes between the anions of the RTILs and the EDs. Using time resolved transient absorption spectroscopy from fs to ns, we will explain the processes following the CT excitation and discuss the species involved. Based on this data we will show that the CT absorption does not only open a pathway for RTIL photo-degradation, relevant in e.g. dye sensitized solar cells, but can also lead to data misinterpretations in photoinduced bimolecular quenching experiments.
[1] Castner Jr., E. W., Margulis, C. J., Maroncelli, M. & Wishart, J. F., Annu. Rev. Phys. Chem., 2011, 62, 85–105.
[2] Koch, M., Rosspeintner, A., Angulo, G. & Vauthey, E., J. Am. Chem. Soc., 2012, 134, 3729–3736.