Direct catalytic oxidation of methane to methanol: Methanol protection using phosphoric acid
The selective partial oxidation of methane to methanol is a difficult yet rewarding task as it has the potential to eliminate the prevalent natural gas flaring by providing novel routes to its valorisation. Despite the emerging number of disparate approaches for this reaction, none has been commercialized yet due to the low product yields as well as a host of engineering issues. A diligent analysis of the large body of work in this field reveals the failure of ‘direct methane to methanol’ (DMTM) systems to reach high yield (Fig. 1a)[1]. Furthermore, the better-performing ones incorporate specific measures to stabilize or derivatise methanol[2]. Such a protection of methanol enables these systems to overcome the rapid fall in product selectivity with increasing methane conversion: a thermodynamic issue that plagues the unprotected DMTM routes.
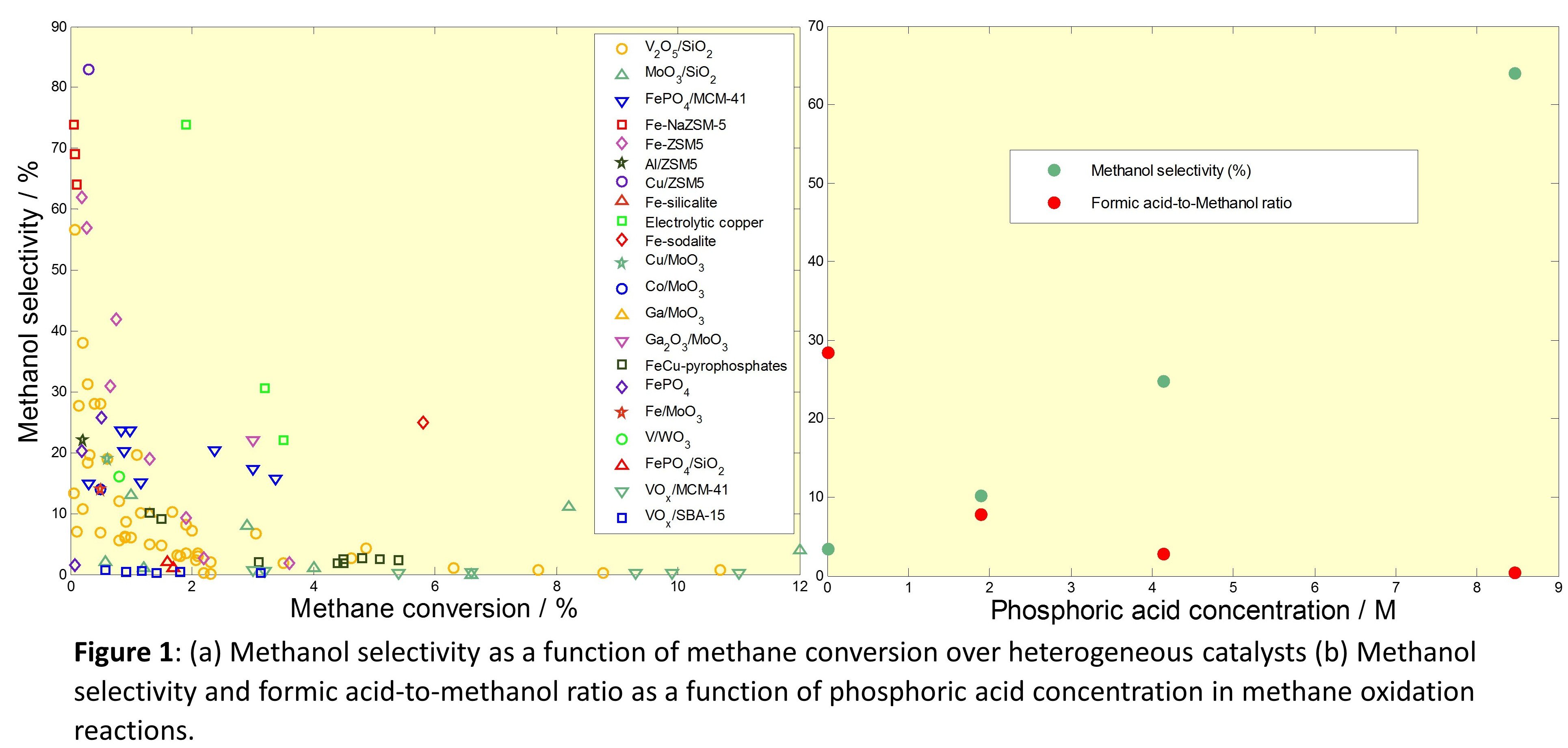
Iron-containing zeolites catalyze the conversion of methane to methanol and a number of other products with aqueous hydrogen peroxide as the oxidant[3]. Formic acid is the major product, formed by the over-oxidation of methanol. The current work shows the possibility of making this process methanol-selective using phosphoric acid. The addition of phosphoric acid to the reaction mixture protects methanol by suppressing its undesired over-oxidation to formic acid. This approach using phosphoric acid is characterized by a much higher methanol selectivity and a higher methanol concentration in solution (Fig. 1b). Furthermore, the efficacy of phosphoric acid is unique, as the use of other acids did not yield propitious results.
[1] M. Ravi, M. Ranocchiari, J. A. van Bokhoven, 2017, submitted.
[2] M. Ahlquist, R. J. Nielsen, R. A. Periana, W. A. Goddard III, Journal of the American Chemical Society 2009, 131, 17110-17115; R. A. Periana, D. J. Taube, S. Gamble, H. Taube, T. Satoh, H. Fujii, Science 1998, 280, 560-564.
[3] C. Hammond, M. M. Forde, A. Rahim, M. Hasbi, A. Thetford, Q. He, R. L. Jenkins, N. Dimitratos, J. A. Lopez‐Sanchez, N. F. Dummer, Angewandte Chemie International Edition 2012, 51, 5129-5133