Light-driven electron accumulation in a molecular pentad
with potential inversion
Charge accumulation plays a crucial role in the greater context of artificial photosynthesis, because carbon dioxide reduction or water splitting rely on multi-electron chemistry. Due to the fact that the second reduction potential is much less negative in comparison to the first, dibenzo[1,2]dithiin is an interesting molecular unit to accumulate two electrons [1]. We expect that the potential inversion could facilitate electron accumulation compared to previously investigated systems [2, 3].
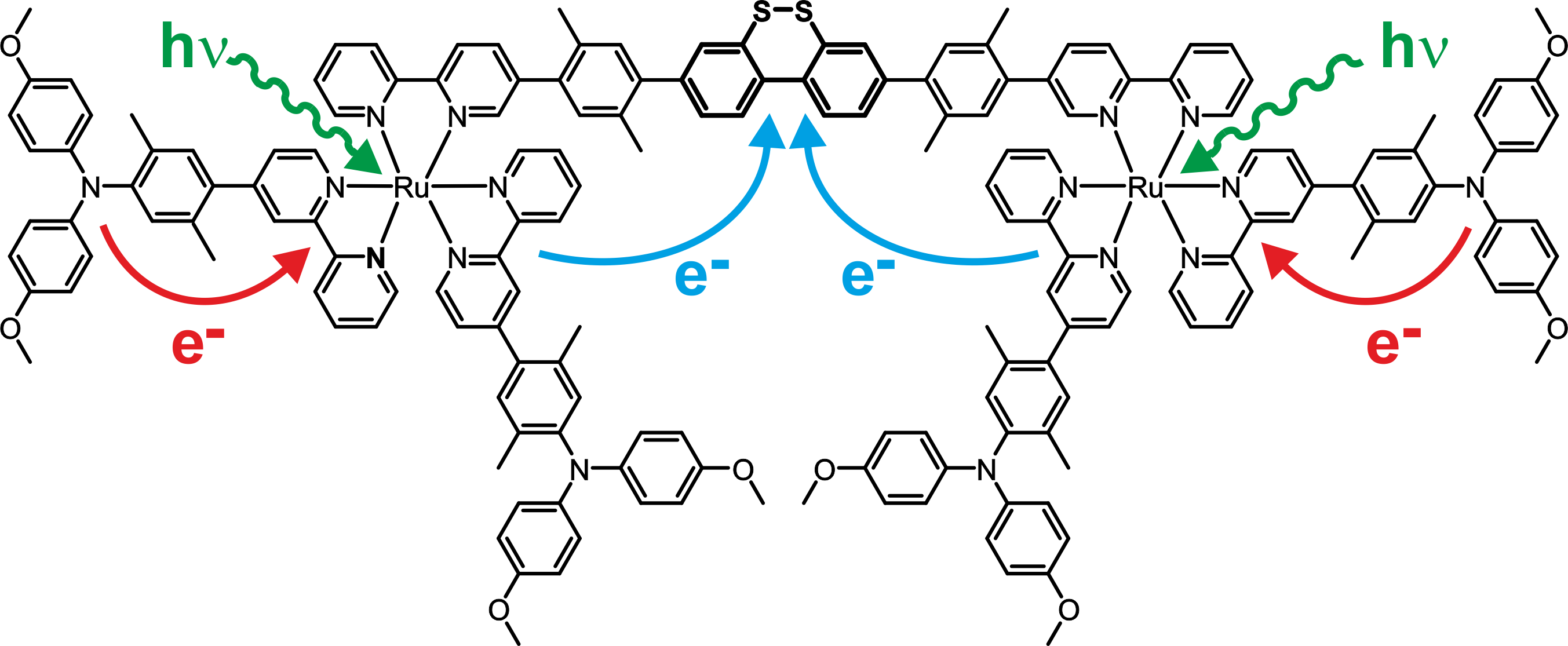
In this work, light-induced charge accumulation on dibenzo[1,2]dithiin was achieved in absence of sacrificial donors. We were able to show that after excitation of the sensitizers, the intramolecular two electron transfer from the triarylamine moieties to the dibenzo[1,2]dithiin unit indeed takes place. In presence of acid, a very long living dithiol species is formed.
[1] Gabriel B. Hall, Rudresha Kottani, Greg A. N. Felton, Takuhei Yamamoto, Dennis H. Evans, Richard S. Glass, Dennis L. Lichtenberger, Journal of the American Chemical Society, 2014, 136, 4012-4018.
[2] Martin Kuss-Petermann, Margherita Orazietti, Markus Neuburger, Peter Hamm, Oliver S. Wenger, Journal of the American Chemical Society, 2017, 139, 5225-5232.
[3] Yann Pellegrin, Fabrice Odobel, Coordination Chemistry Reviews, 2011, 255, 2578-2593.